Research Area # 1: Mitral Valve Disease - Mechanisms & Therapies
|
Project # 1: Mechanobiology of mitral valve prolapse:
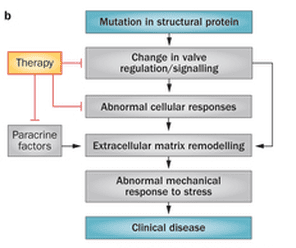
Mitral valve prolapse (MVP) is a common valvular lesion that affects 2-5% of the general population, but its pathogenic mechanisms remain unknown. Though genetic mutations are increasingly reported in patients with MVP, the degenerative pathology of the valve does not manifest until the 4th decade of life or later. Our lab is studying the hypothesis that " genetic mutations lead to breakdown of key mechanosensitive pathways, which inhibit physiological force transduction that is required for normal maturation of the valve." We use pathological specimens from humans, primary valve cell cultures and more recently iPSC derived valve progenitors to define the physiological mitral valve maturation process, so we can investigate MVP mechanisms. |
|
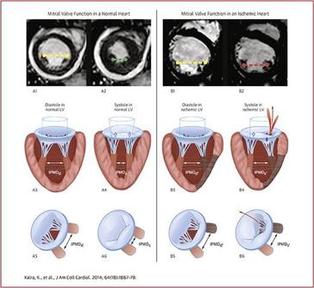
Project # 2: Improving surgical repair of ischemic mitral regurgitation: Ischemic mitral regurgitation (IMR) is a mitral valve lesion that develops in patients surviving a previous heart attack/myocardial infarction or in those with chronic ischemic cardiomyopathy. Though the mitral valve is biologically normal in these patients, dilatation of the left ventricle perturbs the mitral valve structure and restricts its proper closure. Undersizing mitral annuloplasty (UMA) is the current standard for repair, but its durability is rather poor. We are interested in understanding the biomechanics underlying the failure of UMA, and in translating this knowledge to develop new repairs. Our current focus is on translating papillary muscle approximation (PMA) is an effective and durable technique to repair ischemic mitral regurgitation. |
Project # 3: Mitral leaflet remodeling in response to repair:
|
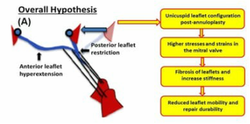
Heart valve leaflets when subjected to chronic, elevated mechanical stretch respond with extracellular matrix synthesis, stiffening and ultimately calcification. Undersizing ring annuloplasty (URA) is a repair that restores mitral valve competence at the expense of leaflet mechanics. URA elevates mechanical stretch on the anterior and posterior mitral leaflets, and can potentially induce fibrosis and calcification. In this project, we seek to investigate if mitral valve leaflets undergo pro-fibrotic remodeling and identify the mechanisms underlying this pathological remodeling. Our hope is to develop targeted therapies that could inhibit this adverse remodeling process.
|